As the pipeline of biologics continues to grow, biopharmaceutical
organizations are outsourcing an increasing number of stability
studies. Careful consideration must be given when choosing a partner for
these programs due to their inherent complexity. Several key points to
consider, including capability, capacity, protocol development, and
project management, are discussed here.
Background
According
to ICh Q1A (R2), “The purpose of stability testing is to provide
evidence on how the quality of a drug substance or drug product varies
with time under the influence of a variety of environmental factors such
as temperature, humidity, and light, and to establish a re-test period
for the drug substance or a shelf life for the drug product and
recommended storage conditions.”
1 In the case of biologics,
ICh Q5C states, “The evaluation of stability may necessitate complex
analytical methodologies. Assays for biological activity, where
applicable, should be part of the pivotal stability studies. Appropriate
physicochemical, biochemical, and immunochemical methods for the
analysis of the molecular entity and the quantitative detection of
degradation products should also be part of the stability program
whenever purity and molecular characteristics of the product permit use
of these methodologies.”
2
ICh provides specific
guidance for biologics for several reasons. When compared to typical
small molecule products, proteins exhibit greater instability, are more
sensitive to the environment, and require more complex analytical
methodologies to fully characterize them. Some of the most common types
of instability are shown in
Table 1.
Table 1.

In
addition, biological activity is highly dependent on interactions (both
non-covalent and covalent), and biologics are particularly sensitive to
environmental factors, such as temperature, light, oxidation, shear,
and ionic content. Therefore, strict storage, packaging, handling, and
transport conditions are usually necessary.
Points to Consider
Validated,
robust, stability-indicating methods are critical to a successful
stability program. As noted above, these studies require quite a wide
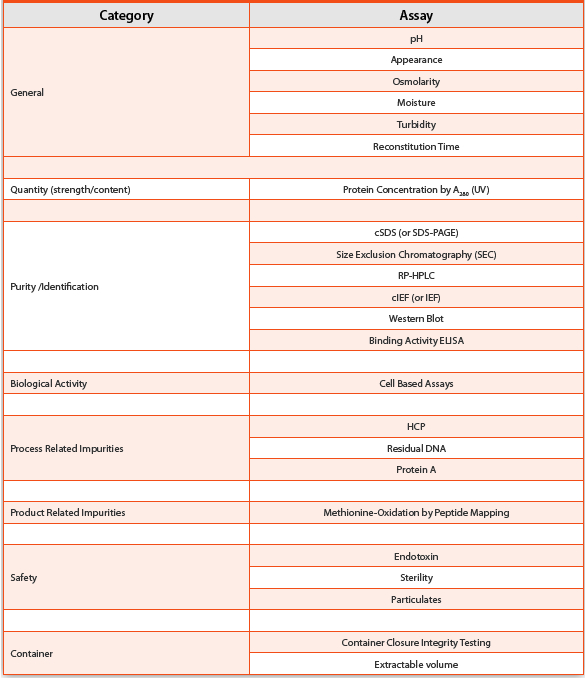
variety of testing and expertise. A typical biologics stability study
may require many of the assays listed in
Table 2. Some
products, such as antibody drug conjugates (ADCs), increase the
complexity immensely as the monoclonal antibody is considered an
intermediate that also needs to be studied. There is a regulatory
expectation that key characteristics linked to critical quality
attributes (CQAs) are measured with orthogonal methods, so multiple
methods may be used to measure purity or other aspects.
Table 2.
Therefore, the laboratory must have the
capability
to perform method installation of a wide array of physiochemical,
microbiological, and biochemical assays. This may involve the transfer
of established methods from the sponsor or the development and
validation of stability-indicating methods (and verification of
compendial methods). These activities and assays encompass a broad range
of skill sets, expertise, equipment, and instrumentation.
The
laboratory should also have the ability to troubleshoot method and/or
product issues and perform investigations of
out-of-specification/out-of-trend results. This is especially important
during early phase stability studies, where unexpected degradation
products and/or contaminants may arise that require further
characterization. Thus, additional, specialized techniques such as
accurate mass (mass spectrometry), micro-flow imaging (MFI), light
scattering, nonroutine lC detection (RI, CAD, and ElSD), and microbial
identification may be necessary.
In conjunction with the capabilities, it is imperative that the laboratory have adequate
capacity.
Stability studies have the potential to produce many samples that need
to be tested in a relatively short time frame. Therefore, redundancy of
specialized equipment and trained staff must be considered.
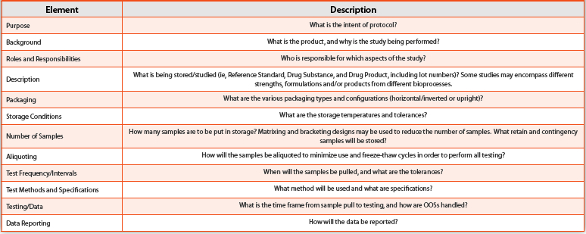
According
to ICh Q5C, the stability protocol should include all necessary
information that demonstrates the stability of the biological product
throughout the proposed expiration dating period. Therefore, clear,
well-written
stability protocols are another vital aspect of
these programs, and the potential outsourcing partner should be adept
and experienced in drafting these protocols. All of this must be
captured in the protocol. Key elements of a stability protocol are
listed in
Table 3.
Table 3.
 Zoom In
Zoom In
Simultaneous
studies are often performed; therefore, careful thought should be given
to finding a balance between batching efficiency and unrealistic sample
volumes. Other considerations such as freeze-thaw logistics should be
covered in the protocol as well. Further, studies performed in support
of development and clinical trials may have different objectives than
those for marketed products, and these differences need to be
considered. Photostability, stress stability, and thermal cycling are
all typically covered in a comprehensive program, and therefore, the
contract organization should have experience with these types of
studies. The development of a stability protocol is ideally carried out
in a collaborative manner between the laboratory and sponsor.
Lastly, as important as the capability, capacity, and stability protocols are to the project, strong
project management
is paramount. large stability programs can be logistically challenging
and require a tremendous amount of planning, forecasting, scheduling,
follow-up, and communication. Regular calls and meetings between the
sponsor and the lab are crucial at least in the lead up and during the
early time points of a study. A well-run meeting should be agenda-driven
and followed up with minutes and action items in a timely manner. Key
discussion topics include: planning for upcoming pulls, sample status,
OOS results, “anomalous” results, investigations, and reference
standards management. A proficient project manager must be able to
effectively interface with the various testing laboratories, Quality
Assurance, stability storage, and the sponsor. Therefore, the laboratory
should have a proven track record in managing biologics stability
programs.
References
- The International Conference on
Harmonisation of Technical Requirements for Registration of
Pharmaceuticals for Human Use. ICH Harmonised Tripartite Guideline.
Stabilty Testing of New Drug Substances and Products Q1A(R2). February
6, 2003. Available at: http://www.ich.org/fi leadmin/ Public_Web_Site/ICH_Products/Guidelines/Quality/ Q1A_R2/Step4/Q1A_R2__Guideline.pdf. Accessed August 27, 2014.
- International
Conference on Harmonisation of Technical Requirements for Registration
of Pharmaceuticals for Human Use. ICH Harmonised Tripartite Guideline.
Quality of Biotechnological Products: Stability Testing of
Biotechnological/ Biological Products Q5C. November 30, 1995



 Zoom In
Zoom In
No comments:
Post a Comment